(The detection of parent geothermal water: Early Showa era, early 20th century)
A young geophysicist, Takaharu Fukutomi (1908-1997) began conducting studies on hot springs during the time when Torahiko Terada began researching the relationship between hot springs and crustal movements. Fukutomi dedicated his life to the establishment of the Department of Geophysics in Hokkaido University's Faculty of Science, educating the younger generation as a professor while simultaneously expanding the field of research on hot springs.
Fukutomi graduated from College of Science, Imperial University of Tokyo, Department of Seismology in 1931. After graduation, he started working at a private laboratory, conducting research on the Izu Peninsula's hot springs. One of his achievements involved the estimation he derived from the correlation between the temperature of Shimokawazu hot spring and the concentration of chloride ions (Figure 1). He presumed that the Shimokawazu hot spring was a mixture of "low-temperature low-concentration groundwater" and "high-temperature high-concentration parent geothermal water", that the temperature of the parental geothermal water was estimated to be above 190°C, with the concentration of chloride ion above 1.0 g/L. (It was first stated as 185°C in the original thesis in 1936, but was amended later.)
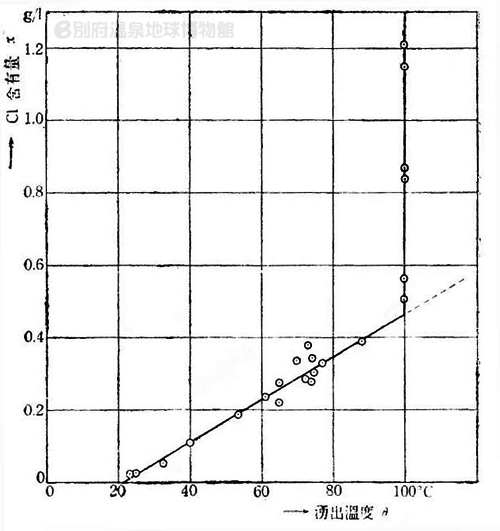
Figure 1: The correlation between the spring temperature (°C)
and the concentration of chloride ion (g/L) in Izu-Shimokawazu hot spring. |
Supplementary comments on the research of Shimokawazu hot spring
There are two types of springs at Shimokawazu, the general spring and the boiling spring. Figure 1 shows a plot of data points from both types of spring, the horizontal axis represents spring temperature values and the vertical axis represents values for the concentration of chloride ion. First, for general springs with a temperature below 100°C, a linear relationship is indicated by a positive slope can be observed between the spring temperature and the concentration. This suggests that the Shimokawazu hot spring is a mixture of "low-temperature low-concentration groundwater" and a "high-temperature high-concentration parent geothermal water." The linear relationship is indicated below:
[concentration (g/L)]=0.0059 × [temperature (°C)] -0.13
Meanwhile, the minimum concentration value of a 100°C boiling spring was approximately 0.5 g/L. All values shown here are higher than this. From this, the underground temperature was estimated to be above 100°C. However, as the high-temperature water rises to the ground surface, the pressure decreases and the water starts to boil. During this process, vaporized heat (latent heat of evaporation) is lost, therefore the temperature is not able to exceed the 100°C boiling point under atmospheric pressure.
On the other hand, the high-temperature water from the boiling spring is transformed into vapor, causing the concentration of chloride ions to increase.
Given the above, Fukutomi proposed that the boiling effect has to be taken into consideration when estimating the concentration and the temperature of underground hot water that exceeds 100°C using the data from the boiling spring.
Incidentally, the underground temperature and the concentration can be calculated using the measurements of both the hot water in liquid form and the vapor yielded.
The linear relationship between spring temperature and the concentration of chloride ion in the Kamisuwa hot spring.
In 1932, Shinkichi Yoshimura (1906-1947) had identified the linear relationship between the spring temperature and the concentration of chloride ion at Kamisuwa hot spring in Nagano prefecture, even before the data from Shimokawazu hot spring was obtained.
Shinkichi Yoshimura was a limnologist and a geographer, well known for his contributions to the studies of limnology by developing limnology studies in Japan to a world-class level. Yoshimura’s energy appeared to be inexhaustible. He published hundreds of journals and papers, writing the highly acclaimed Great Book of Limnology. Although his research on hot springs is relatively unknown, the data on Kamisuwa hot spring, which he published during the early period of his research, is said to be monumental.
While accomplishing great achievements as a researcher, he became a meteorological technician at the Central Meteorological Observatory (currently the Japan Meteorological Agency). On January 21st, 1947, Yoshimura died during an observation at Lake Suwa due to cracked ice. He was only 41.
The data shown in Figure 1 was also obtained at Kamegawa hot spring in Beppu (Figure 2). The temperature of parenr geothermal water in Kamegawa hot spring was estimated at above 160°C, with the concentration of chloride ion at 2300mg/L (1966).
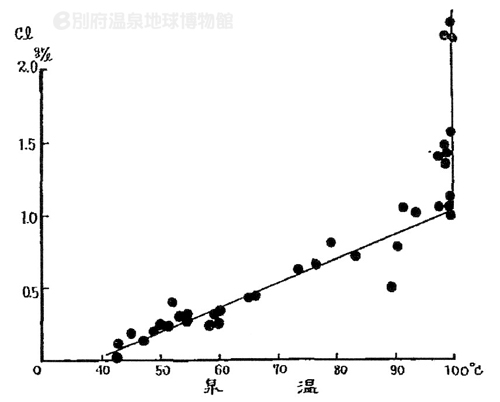
Figure 2: The relationship between the spring temperature
and the concentration of chloride ions in Kamegawa hot spring |
(Additional Information)
Professor Takaharu Fukutomi had a close relation with Oita Prefecture. Although he was born in Nagasaki Prefecture, he moved to Oita when his father relocated for work, and graduated from Kitsuki Middle School (currently, Kitsuki High School) in Kitsuki City, Oita Prefecture. His research covers a broad range of fields such as seismology, volcanology, tectonism, oceanography, limnology, and hot spring science. He left behind 66 scientific papers on hot springs, and I would like to refer them here occasionally. Additionally, The Physics of Hot Springs, which I frequently cited, was written by Prof. Fukutomi when he was 28.
Takaharu Fukutomi (1936). The Physics of Hot Springs. Iwanami Shoten.
Kozo Yuhara & Kinzo Seno (1969). Geosience of Hot and Mineral Springs. Chijin Shokan.
Kosaburo Yamashita & Tadayoshi Mori (1966). Water System of Kamegawa Onsen in Beppu City, Reports of Oita Prefecture Hot Springs Research Society, No. 17, 12-17.
↑TOP